Plastics are ubiquitous, and their diversity is almost impossible to fully capture. Due to their versatility, this indispensable material is used across nearly all industries and sectors: from packaging and construction to transportation, electrical and electronics industries, agriculture, medicine and sports, as well as in households and clothing, adhesives, and paints. But how are plastics made – and what does the plastic manufacturing process actually involve?
Basics: What Are Plastics?
“Plastic” is a widely used umbrella term for an entire class of materials. It refers to synthetic or semi-synthetic materials composed of macromolecules. In technical terminology, these are called polymer molecules or simply polymers. However, products made from them – such as hoses, seals, or drive components – can differ significantly in their properties, because not all plastics are the same.
They can consist of different chemical compounds and are derived from various sources. Some monomers are produced naturally, others synthetically. The simplest type of monomer is alkenes – compounds from the group of aliphatic hydrocarbons containing a -C=C- double bond at any position in the molecule. Examples include ethylene (C2H4), propylene (C3H6), and butene (C4H8).
Functional Groups and Additives in Plastics
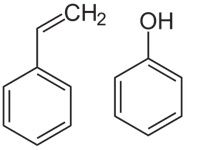
Monomers may contain functional groups, meaning chemical groups attached to a molecule that impart specific properties. Examples include styrene (C8H8), which contains a vinyl group (-CH=CH2), and phenol (C6H5OH), which carries a hydroxyl group (-OH).
However, the final properties of a plastic product are not determined solely by the polymers themselves but also by additives. These are auxiliary substances such as plasticizers, colorants (pigments), fillers, reinforcing agents, or stabilizers. They can be added before, during, or after polymerization and allow targeted modification of the plastic’s properties.
Raw Materials: Fossil Fuels and Bio-Based Plastics
The raw materials used in plastic manufacturing are fossil fuels, most commonly crude oil. This complex mixture of hydrocarbons is separated into fractions in a refinery through distillation and subsequently undergoes cracking. In this process, larger hydrocarbon molecules are broken down into smaller molecules – monomers – by applying heat and pressure in the presence of a catalyst.
Bio-based plastic manufacturing relies on renewable plant-based resources such as corn, sugarcane, or soybeans, as well as plant residues or waste materials. The milky sap of the rubber tree, known as latex, is another example and forms the basis for many products made from natural rubber, such as rubber hoses and seals.
Depending on the nature of the raw material and the desired properties of the bio-based material, the components are extracted. This can be achieved through fermentation, where microorganisms break down plant sugars or carbohydrates into simpler molecules such as ethanol or lactic acid. Another method involves chemical conversion using catalysts or other substances. In both cases, the resulting monomers are subsequently polymerized.
Manufacturing: Polymerization of Plastics
One of the most common methods in plastic manufacturing is chain-growth polymerization, which enables the production of a wide range of materials with diverse properties. It proceeds in several stages: In the first step, initiation, free radicals are generated by light, heat, or chemical reactions – atoms with an unpaired valence electron. This creates a reactive site in the monomer for bonding with another monomer. In the second step, propagation, the monomer reacts with others to form a growing chain. This chain continues to grow by adding further monomers. In the subsequent chain transfer stage, the free radical may transfer to another molecule, initiating a new polymer chain. In the final step, termination, polymerization is stopped – either by collisions between polymer chains or by lowering the reaction temperature. This process is commonly used to produce thermoplastics such as polyethylene (PE), polystyrene (PS), or polyvinyl chloride (PVC).
In polycondensation, two or more monomers react while releasing by-products such as water or methanol. Similar to chain-growth polymerization, the process occurs in multiple stages. During initiation, a functional group of a monomer is activated – for example by adding a catalyst. In the propagation phase, it reacts with the functional group of another monomer, forming a polymer chain that continues to grow. In the final step, termination, polymerization is halted, for instance by chain collisions or inhibitors. Examples include polyesters (PES), polyamides (PA), and polyurethane (PUR).
Polyaddition is a polymerization method in which no by-products are formed. It proceeds in two stages. First, a functional group of a monomer is activated, for example by a catalyst. The activated group then reacts with functional groups of other monomers. The resulting polymer chain continues to grow until no reactive groups remain and all monomers are bonded. This method is used to produce materials such as epoxy resins or polycarbonate (PC).
Spatial Cross-Linking of Polymer Chains
During polymerization, different structures can form. In linear polymerization, monomers are linked in a chain without cross-links between chains. This can occur through chain-growth polymerization or polycondensation.
In cross-linked polymerization, chemical bonds form a three-dimensional network structure between polymer chains. This is achieved through reactions between reactive groups or by adding cross-linking agents, typically in polyaddition processes. In some cases, partial cross-linking occurs, resulting in a combination of linear and network structures.
The choice between linear and cross-linked polymerization depends on the desired properties and application requirements. Linear polymers may offer flexibility, elasticity, and recyclability, whereas cross-linked polymers provide higher mechanical strength, dimensional stability, and resistance to environmental influences.
Further Processing in Plastic Manufacturing
After polymerization and the addition of any additives, the next stage of plastic manufacturing involves shaping polymers into their final form using appropriate processes. Among the many available methods, the most important are outlined here: In extrusion, the heated polymer is forced through a die to form shapes such as plastic hoses or profiles.
Injection molding is frequently used for items such as toys, household goods, or technical components, where molten polymer is injected into a mold. Cold forming shapes polymers at room temperature into films or sheets. In blow molding, the polymer is melted and then inflated into a mold, producing items such as bottles.
What Types of Plastics Are There?
Polymers can be classified according to various criteria. One key classification is based on physicochemical properties.
Thermoplastics
Thermoplastics consist of linear polymer chains that are not chemically cross-linked into a rigid three-dimensional network. Instead, they are held together by intermolecular forces such as van der Waals forces, which can be overcome with sufficient heat.
This allows thermoplastics to be melted and reshaped repeatedly without altering their chemical structure. The process is reversible and enables recycling. At the same time, thermoplastics can exhibit high strength and chemical resistance. Examples include polyethylene (PE), polypropylene (PP), polystyrene (PS), and polyvinyl chloride (PVC).
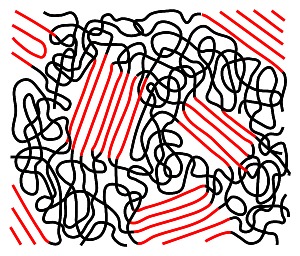
Thermoplastics are widely used in the manufacturing of packaging, household goods, and technical components. Common end products made from polyethylene include plastic foils, containers for liquids, as well as hoses and adhesive tapes. The more rigid and thermally resistant polypropylene is often used for applications such as filter meshes or plastic screws. Lightweight polystyrene is commonly used for test tubes and cuvettes, while PVC is frequently used for hoses and semi-finished products.
Elastomers
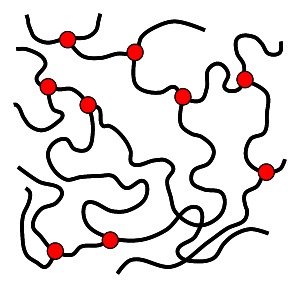
Elastomers consist of loosely cross-linked, three-dimensional macromolecules. The cross-linking usually occurs through covalent bonds between polymer chains. These strong bonds prevent the chains from separating under normal temperature and pressure conditions, which is why elastomers are generally not meltable.
When subjected to external force, the polymer chains can be stretched reversibly. Once the force is removed, they return to their original position, resulting in elastic behavior. Examples include natural rubber (latex) and synthetic elastomers such as polyisoprene or polychloroprene (neoprene). Elastomers are widely used in the manufacturing of tires, seals, hoses, sports shoes, O-rings, and more.
Thermosets
Thermosets are highly cross-linked materials that are rigid and non-elastic compared to elastomers. Their tightly cross-linked polymer chains form a stable, non-thermoplastic structure. The cross-linking occurs through strong covalent bonds, forming a highly cross-linked three-dimensional network.
Thermosets are also an important part of everyday life. Materials such as polyesters, epoxy resins, or formaldehyde resins are used in the manufacturing of furniture, doors, electronic components, housings, or gears. Epoxy resins, for example, offer excellent mechanical properties and resistance to temperature and chemicals. Their applications range from structural adhesives to medium-voltage insulators.
Sustainability of Plastics and Disposal
Due to differences in raw materials and manufacturing processes, plastics vary significantly in terms of biodegradability and recyclability. Conventional fossil-based materials are difficult to biodegrade – if at all – and contribute to plastic waste and microplastic pollution. Recycling is complex due to their composition.
They can also be used for energy generation through incineration. However, this approach is not sustainable, as it releases pollutants and contributes to climate change.
Responsible use and proper handling of plastic materials crucial for sustainability and environmental protection. A key measure is reducing overall waste volumes – by avoiding single-use products and promoting reusable solutions.
However, less than 20 % of plastic waste in Germany is actually recycled. For these materials to become valuable resources, they must be integrated into a circular economy. The transition requires significant changes in how products are designed, used, and recovered. The German plastics industry has committed to this transformation.
Solutions must be found to reduce reliance on crude oil while making plastic manufacturing more sustainable. Governments, businesses, and consumers must collaborate to improve the sustainability of these materials and develop innovative methods for efficient recycling and waste management.
Image sources: Feature image | © molekuul.be - stock.adobe.com Structure of styrene: By NEUROtiker - Own work, public domain, https://commons.wikimedia.org/w/index.php?curid=3223780 Structure of phenol: By NEUROtiker - Own work, public domain, https://commons.wikimedia.org/w/index.php?curid=3223795 Polymer structure, semi-crystalline: By Roland.chem - Own work, CC0, https://commons.wikimedia.org/w/index.php?curid=37866505 Loosely cross-linked polymer: By Roland.chem - Own work, CC0, https://commons.wikimedia.org/w/index.php?curid=37866466 Densely cross-linked polymer: By Roland.chem - Own work, CC0, https://commons.wikimedia.org/w/index.php?curid=37866493
 Reichelt Chemietechnik Magazine
Reichelt Chemietechnik Magazine