With a share of 6% of the total amount of all plastics produced, polyethylene terephthalate, or PET for short, is one of the most important representatives. Originally developed for fiber applications, polyethylene terephthalate has become an important everyday plastic.
In addition to beverage bottles, packaging, outdoor and sports clothing, seat belts, blankets, and tents contain this polymer. PET is also often used as a carrier material for films, adhesive tapes and filter cloths, as well as in the form of functional components such as gears or pump parts.
History of PET Plastic
The Second World War slowed further research and development work on this new plastic. In 1947, the British chemical group Imperial Chemical Industries (ICI) received the worldwide patents for it, except for the USA. Two years later, ICI started trial production of fibers made from polyethylene terephthalate under the brand name TERYLENE®. The US chemical company DuPont acquired the manufacturing rights for the USA and produced PET fibers under the trade name DACRON®.
After licenses were granted to the German companies Farbwerke Hoechst AG and Vereinigte Glanzstoff-Fabriken AG in Wuppertal, PET was also produced in Germany from 1956 under the names Trevira® and DIOLEN®.
Production of PET
Starting materials for the production of polyethylene terephthalate are terephthalic acid, correctly referred to as 1,4-benzenedicarboxylic acid, and ethane-1,2-diol with the trivial name ethylene glycol, as shown in the following structural scheme, which is intended to illustrate the chemical structure.
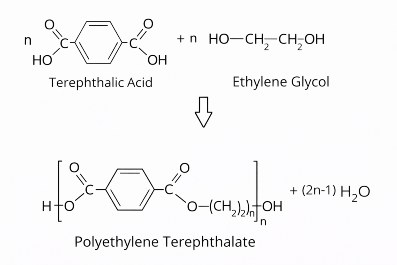
In the first step, the monomers are directly esterified at temperatures between +220 °C and +260 °C to form a dimeric intermediate. In the subsequent polycondensation, this intermediate forms the polymer PET, with water as a byproduct.
The polycondensate is present as a viscous melt that is pressed into thin threads, ribbons, or strands. After cooling, the material can be further processed into granules.
Properties of Polyethylene Terephthalate
Polyethylene terephthalate belongs to the group of polyesters. These are thermoplastics that can be reversibly deformed within a certain temperature range. Due to the ester group in the main chain, PET is polar; the polymer consists of linear chains without cross-linking.
Density and Temperature Resistance
The properties depend on the degree of crystallization. The density of PET-A is between 1.33 and 1.35 g/cm3, and that of PET-C between 1.38 and 1.40 g/cm3. PET-C has higher stiffness and hardness, but lower impact strength than PET-A.
The maximum continuous service temperature of semi-crystalline polyethylene terephthalate is +100 °C, and the minimum is -20 °C. For amorphous PET-A, values of +60 °C and -40 °C are given.
Glass Transition Temperature of PET
PET-C has a glass transition temperature of +98 °C, PET-A of +86 °C. The glass transition temperature Tg is the temperature at which an amorphous or partially amorphous material changes from a rigid, glass-like state to a soft, rubber-like one. Specifying the glass transition temperature is important for processing thermoplastics.
Electrical Resistance and Chemical Resistance
Polyethylene terephthalate has a high specific resistance, high dielectric strength, and a high dielectric constant. It has very low sliding friction, shows little shrinkage, and therefore offers high dimensional stability.
The polymer is resistant to weak acids and alkalis, fats, oils, aliphatic and aromatic hydrocarbons. PET is attacked by strong acids and alkalis, halogenated hydrocarbons, and prolonged contact with hot water. Although the material is combustible, it is difficult to ignite according to UL 94 after the “tests for the flammability of plastics for parts in devices and applications”.
What Is PET Used for?
PET-A as a Material for Beverage Bottles and Films
PET-A is characterized by good light transmission. The best-known application of this polymer is beverage bottles. Compared to glass, the plastic is lighter and more shatter-resistant. A 1-liter single-use PET bottle weighs between 20 and 30 g; a refillable PET bottle around 100 g. A glass bottle, by contrast, weighs around 600 g. The lower weight of these bottles also has an impact on transport costs and CO2 emissions.

The plastic bottles are manufactured in a two-stage process. In the first step, the PET granulate is melted at a temperature of around +280 °C and, by injection molding, so-called preforms are produced. The preforms already have a thread. In the second step—usually carried out directly before filling—the preforms are heated to around +120 °C in a stretch blow-molding process and blown into bottles using sterile compressed air. Manufacturing directly before filling, at a higher temperature, simultaneously ensures the sterility of the bottles.
However, PET bottles are also controversial. Antimony trioxide (Sb2O3) is used as a catalyst in the production of the bottles, producing acetaldehyde. Both chemicals can dissolve in small amounts into the liquid filled in PET bottles.
In addition to PET bottles, the amorphous PET variant is also used in the form of foils for packaging foods and pharmaceuticals.
Use of Semi-Crystalline Polyethylene Terephthalate PET-C
PET-C is mainly processed into fibers for the textile industry or into intermediate products for filtration technology such as screen fabrics, filter cloths, and nonwovens. PET fibers are known under the name “polyester”. These fibers have hardly any stretch, are wrinkle-free, and tear-resistant. Because they absorb very little water, they are often found in sports and outdoor textiles as well as in awnings, tents, sails, and flags. Due to their strength, they are used to manufacture seat belts, airbags, and strapping bands for load securing. As a lofted material, they are used in pillows, blankets, sleeping bags, and jackets.
Due to its high dimensional stability, good sliding and friction behavior, and very good machinability, polyethylene terephthalate is also suitable for components that must be manufactured with tight tolerances, such as vascular prostheses. The polymer is also an integral part of drive technology; for example, it is used to manufacture gears, precision plain bearings, rollers, couplings, and parts for pumps.
The material is also frequently used in industrial and assembly adhesive tapes and is also used for films and filter cloths. Many molded and housing parts for household and office devices are also made from this material.
Recycling of PET
Polyethylene terephthalate can be identified by the recycling code with the digit 1. Thanks in part to Germany’s deposit-return system for single-use beverages, 94% of PET bottles in Germany are recycled.
The URRC process was developed by the American company United Resource Recovery Corporation (URRC). It also starts with washing, followed by a float-sink separation, in which light plastics such as polyethylene (PE) and polypropylene (PP) float on the surface while polyethylene terephthalate sinks to the bottom. After the flakes are dried, light contaminants such as labels are removed using an air separator. The cleaned PET can then be processed into fibers. To obtain food-grade PET, the cleaned flakes are treated with sodium hydroxide solution to remove contaminants and slightly dissolve the flake surface. After further washing and drying steps, the flakes can be filled for further processing.

According to a 2019 study on PET beverage bottle collection and recycling in Germany by the Society for Packaging Market Research (GVM), 94% of all PET bottles in Germany are recycled. One bottle contains an average of 30% recycled material. Refillable bottles can be filled up to 20 times. In addition, PET bottles have a better environmental footprint than glass bottles or aluminum cans, both in production and transport, as well as in recycling. This means that PET bottles are better than their reputation.
Image sources: Featured image | © ximich_natali – stock.adobe.com PET preforms with thread before the stretch blow-molding process | © Pixel_B – stock.adobe.com PET recycling symbol on a beverage bottle | © calypso77 – stock.adobe.com
 Reichelt Chemietechnik Magazine
Reichelt Chemietechnik Magazine






