Physical processes and chemical reactions often exhibit a significant dependence on temperature. Electrical resistance, as described by Ohm’s law, increases with rising temperature in metallic conductors, whereas it decreases with increasing temperature in semiconductors. Liquid mixtures are separated by distillation, in which heating causes the individual components to evaporate sequentially according to their boiling points and subsequently condense again through cooling. The reaction rate – temperature rule (RGT rule) according to Jacobus Henricus van’t Hoff (1852-1911) states that the rate of chemical reactions approximately doubles to quadruples with an increase in temperature of 10 Kelvin. Heating and cooling equipment is therefore essential for any laboratory.
These examples clearly demonstrate the importance of heating and cooling equipment in the laboratory. The comparability of physical and chemical experiments cannot be ensured without specifying a constant temperature.
Requirements for Heating and Cooling Equipment
Another important factor is control accuracy, i.e. how precisely temperature changes and setpoints approaches can be regulated. Since both heating and cooling require energy, cost is also a significant consideration.
Safety aspects must not be overlooked in order to prevent personal injury and property damage. For heating flammable liquids, for example for distillation purposes, explosion-proof heat sources are suitable as they prevent the ignition of explosive substances. This is referred to as explosion protection (EX protection).
It must also be determined whether heating or cooling should be carried out directly or indirectly. Direct methods include heating by flame or cooling using an ice/water mixture or liquid nitrogen. Indirect methods involve, for example, a cooling or heat transfer medium flowing through a heat exchanger in the form of a coil, which is located within the medium to be cooled. A clear example is the radiator in a car: coolant heated by the engine flows through the heat exchanger at the front of the vehicle, where it is cooled by ambient air. As a result, the coolant itself is cooled as it passes through the heat exchanger.
Indirect heat exchange in the laboratory can be carried out using heat exchanger hoses equipped with a flat profile on one side. This allows reaction flasks, measuring instruments, beakers, chromatography columns, and other laboratory vessels to be temperature-controlled indirectly.
Heating Equipment in the Laboratory – An Overview
Heating in the laboratory can be achieved using fossil fuels, typically propane or butane gas, electrically, or by means of an electromagnetic alternating field.
Gas Burners
Gas burners, such as the Bunsen burner or its technical variants – the Teclu burner, Meker burner, and blast burner – can quickly reach relatively high target temperatures. However, temperature control via the air supply is limited. Gas is supplied through the characteristic red chloroprene rubber gas hoses. Open flames are unsuitable as heat sources for flammable substances, as well as for flammable heat transfer media.
Ovens and Drying Cabinets
Ovens are available in numerous designs with regard to size, temperature range, and control technology. They are typically used to maintain higher temperatures over extended periods with relatively low control deviations. Good thermal insulation from the surroundings is essential for this purpose. As oven size increases, so does thermal inertia – the delay with which an oven responds to control deviations. In addition, the time required to reach the target temperature increases accordingly.
Heating, drying, or warming cabinets are available in various sizes for temperature ranges up to approximately +250 °C (+482 °F). Their design resembles that of a kitchen oven, with or without integrated ventilation. Due to their low temperature deviations and thermal inertia, drying cabinets are well suited to tempering or drying equipment and materials. If flammable gases are produced during use, these may form explosive mixtures with air. The release of toxic substances in the form of gases or dust also presents a potential hazard.
Electric Heating Blowers and Heating Mantles
Electric heating blowers are a practical alternative to gas flames. Smaller quantities of substances can be heated in a short time, with temperatures ranging from +250 to +500 °C depending on the device. Since the airstream is heated by glowing coils, heating blowers are likewise unsuitable for flammable and easily ignitable substances. Only coarse temperature control is possible. Heating blowers also retain considerable residual heat after being switched off and can therefore act as ignition sources.

The geometry of a heating mantle is adapted to the shape of the vessel it partially encloses. It has an inner lining made of fire-resistant material, such as glass fiber, into which the electric heating element is integrated. Laboratory heating mantles are used to heat laboratory glassware such as round-bottom flasks efficiently, evenly, and with minimal deviation from the target temperature. From a safety perspective, heating mantles are also a good choice and are frequently used in school laboratories and academic settings. Larger versions serve as industrial heating mantles in industrial processes.
Microwave Devices
Microwave devices are also used in laboratory applications. An entire field of synthesis chemistry has developed around microwave-initiated reactions, which can lead to entirely new reaction pathways. Microwave-assisted digestion techniques have likewise been an established method in analytical chemistry for over 30 years. A key advantage is the simplicity of the process, analogous to domestic microwave use for rapidly heating food. However, when using closed systems in microwave ovens, safety considerations play a more significant role in laboratory settings than in domestic use.
With additional time programming, it is possible to define different temperatures with specific holding times over any desired period.
Cooling Equipment in the Laboratory
Dewar Vessels and Cooling Baths
Cooling in the laboratory is often carried out using open systems such as cooling baths. These consist of thermally insulated containers whose heat exchange with the surroundings is minimized by a vacuum-insulated double-walled glass jacket with a reflective coating. These Dewar vessels, named after the Scottish physicochemist James Dewar (1842-1923), contain cooled liquids or cooling mixtures. Equipment to be cooled is immersed directly in the vessel.
Coolants and cooling mixtures are selected based on the target temperature and safety considerations. An ice-water mixture ensures a temperature of 0 °C (32 °F) as long as both ice and water are present simultaneously. Adding sodium chloride lowers the temperature to approximately -17 °C (1 °F). A dry ice-ethanol mixture – solid carbon dioxide dissolved in alcohol – can reach temperatures of up to -72 °C (-98 °F).
Closed Cooling Systems
In addition to open cooling baths, closed cooling systems are also used in laboratories. A modern example is the Peltier element, named after the French physicist Jean Peltier (1785-1845): an electrothermal converter that generates a temperature difference when an electric current flows through it. It is also familiar from portable cooling boxes connected to a 12 V vehicle battery. Peltier elements are well suited to applications where only a modest temperature difference relative to the surroundings is required. Their high power consumption, however, limits their use. The compact design can be miniaturized, making this equipment suitable for temperature control of measuring instruments as well.
Laboratory Refrigerators and Cryostats
Laboratory refrigerators, comparable to household appliances, are also widely used. Cooling is achieved through a circulating refrigerant in a closed system. The refrigerant evaporates inside the unit and condenses outside under pressure. The heat required for evaporation is extracted from the interior, resulting in cooling.
Refrigerators in Labs are suitable for storing volatile or temperature-sensitive substances. Uniform temperature distribution within the equipment is important. Depending on the application, specialized versions are available for storing pharmaceuticals, blood products, or – in explosion-proof configurations – flammable substances. Climate chambers, which are programmable to maintain defined temperature and humidity conditions, are also noteworthy. They are used for corrosion testing, as incubators for biological samples, or for accelerated material aging studies.
Cryostats
A special variant is the cryostat, designed to maintain very low temperatures over extended periods. Several system types exist. In bath cryostats, the element to be cooled is surrounded by a cryogenic liquid. Liquid nitrogen, with a boiling point of -195.8 °C (-320.4 °F), or liquid helium, boiling at -268.9 °C (-452.0 °F), are commonly employed.

Evaporation cryostats function similarly to refrigerators. Cold gas generated by evaporating a cryogenic liquid provides the cooling effect, enabling a wide temperature range to be covered. For smaller applications with lower energy consumption, refrigerator cryostats are available. These operate without cryogenic liquid, using a miniature refrigeration unit based on the Linde principle, named after the German engineer Carl von Linde (1842-1934). As compressed gas expands through a nozzle, a temperature drop occurs. This Joule-Thomson effect – named after British physicists James Prescott Joule (1818-1889) and William Thomson (1824-1907) – can be felt when applying aerosol sprays to the skin, as the compressed propellant gas cools upon release.
With dilution cryostats, temperatures just a few thousandths of a degree above absolute zero (-273.15 °C/-459.67 °F or 0 K) can be achieved. Cooling is accomplished by exploiting the enthalpy of mixing of two helium isotopes.
Cryostats find application in materials science, medicine, and biology – for example, to produce ultrathin sections (microtome slices), to cool radiation sensors, or to cool superconducting coils.
Heat Transfer Media in Open and Closed Systems
Direct and Indirect Heat Transfer
Heat transfer media transport thermal energy from a region of higher temperature to one of lower temperature. Depending on the temperature range and application, they are classified as heating media, cooling media, or – below 0 °C (32 °F) – refrigerants. A heating medium conveys heat to a laboratory system at a lower temperature, raising it to the desired level. Conversely, a cooling medium or refrigerant extracts heat from a system, lowering its temperature. Heat transfer media should ideally exhibit high specific heat capacity, a large heat transfer coefficient, and high thermal conductivity, and should be non-toxic, non-flammable, and non-corrosive towards the materials in contact. When used as a heating medium, a high boiling point is desirable; when used as a cooling medium or refrigerant, a low freezing point is essential.
Direct heat transfer involves the simultaneous transfer of heat and matter. The simplest example is adding ice directly to a substance or mixture. Indirect heat transfer means that the medium to be tempered is separated from the heat transfer medium by a thermally conductive barrier. This is typically achieved using coiled pipes or hoses that surround or are immersed in the target system. The two media remain separated by the wall of the hose or plastic tube, preventing any mixing. Gases such as air or nitrogen are also employed as heat transfer media in laboratory blowers.
Common Heat Transfer Media
They differ in their usable temperature range and safety characteristics. Water is a liquid between 0 °C (32 °F) and +100 °C (212 °F), which defines its application range.
By adding PEG, the freezing point can be lowered, enabling cooling well below 0 °C (32 °F) with water/PEG mixtures. Thermal oils are available in natural and synthetic forms. Natural thermal oils include mineral and vegetable oils; synthetic thermal oils include silicone oils. The latter are particularly popular in laboratory work due to their high chemical stability, physiological inertness, and reusability. Sand also serves as a solid heat transfer medium: the reaction vessel is placed in a sand bath heated by a hot plate or open flame.
Laboratory Condensers
Laboratory condensers, typically made of glass, are used to condense gaseous substances and are among the most commonly used devices in laboratories. Depending on the application, a distinction is made between product condensers and reflux condensers.
Product Condensers and Reflux Condensers
Product condensers are installed at a downward angle so that the condensate flows into a collection vessel. Reflux condensers, by contrast, are mounted vertically above the reaction vessel so that condensed vapor is returned to it. Examples of product condensers include the Liebig condenser, named after the German chemist Justus von Liebig (1803-1873), and the coil condenser. A classic reflux condenser is the Dimroth condenser, named after the German chemist Otto Dimroth (1872-1940).

Water is the most commonly used indirect cooling medium, supplied to and discharged from the condenser via rubber hoses. For improved cooling of low-boiling substances, the water can be pre-cooled in an upstream thermostat before entering the condenser. For boiling points above approximately +160 °C (320 °F), air condensers are also used. The air condenser is the simplest type: a straight glass tube used vertically as a reflux condenser or inclined as a product condenser, cooled by ambient air in both cases.
Cooling Performance and Operating Principle
Cooling performance can be improved by increasing the surface area of the condenser. A good example is the bulb condenser, which features periodic constrictions that enlarge the cooling surface – together with the resulting vapor turbulence, this significantly enhances efficiency. Both the coil condenser and the Dimroth condenser achieve higher cooling performance by the same principle: an enlarged surface area combined with induced vapor turbulence.
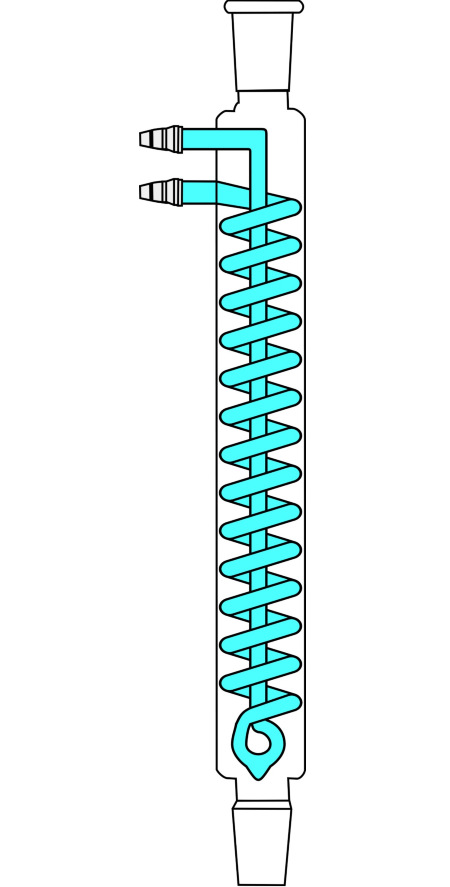
Condensers generally operate on the countercurrent principle, in which hot and cold media flow past each other from opposite ends. This must be observed when connecting the inlet and outlet hoses for the cooling medium.
Important Safety Aspects
Numerous incidents can arise from improper handling of heating and cooling equipment in the laboratory. Even straightforward situations – such as bumping, the sudden violent boiling of a liquid in the absence of suitable nucleation aids – can cause accidents.
Fume hoods prevent substances from the reaction or cooling media from entering the laboratory atmosphere. The use of technical equipment requires adequate expertise. A thorough understanding of the physicochemical properties, toxicity, and reactivity of the substances and materials involved is essential. Emergency procedures are generally set out in laboratory regulations.
Electrical installations are equipped with appropriate safety devices so that even untrained personnel can shut them down in an emergency. All thermal laboratory systems in continuous operation are clearly identified and display instructions for emergencies as well as the relevant contact persons.
Medical emergency equipment and first aid supplies are available in every laboratory area, along with contact details for medical assistance.
Image sources: Feature image | © zyabich – stock.adobe.com Heating mantles | © Sergey Ryzhov – stock.adobe.com Cryostat for histotechnology | © Orlando, CC BY-SA 2.0 <https://creativecommons.org/licenses/by-sa/2.0>, via Wikimedia Commons Distillation setup with Liebig condenser | Distillation_2.jpg: Arlenderivative work: © Mario Link, CC BY 2.0 <https://creativecommons.org/licenses/by/2.0>, via Wikimedia Commons Dimroth reflux condenser | © Sivicia, CC BY-SA 3.0 DE <https://creativecommons.org/licenses/by-sa/3.0/de/deed.en>, via Wikimedia Commons
 Reichelt Chemietechnik Magazine
Reichelt Chemietechnik Magazine






