About 100 years ago, the production of purely synthetic plastics—made exclusively from artificial components—began. In 1907, the Belgian chemist Leo Hendrik Baekeland (1863–1944) developed a process for producing and processing a phenolic resin. He called this plastic Bakelite, and it was the first plastic to be manufactured industrially on a large scale. In 1912, Fritz Klatte (1880–1934) patented a process for producing polyvinyl chloride, better known as PVC.
Polypropylene was first synthesized in 1951 by the Americans J. Paul Hogan (1919–2012) and Robert Banks (1921–1989). In 1953, the German chemist and long-standing director of the Max Planck Institute for Coal Research in Mülheim, Karl Ziegler (1898–1973), developed catalysts for the polymerization of ethene, which had previously been produced at high pressures and temperatures of up to 300 MPa and +300 °C.
In the synthesis, he used organometallic catalysts that made it possible to work at lower pressure and temperature, enabling large-scale industrial use.


In 1954, Karl Rehn succeeded in synthesizing polypropylene at the Hoechst dye works using Ziegler catalysts. At the same time, the Italian chemist Giulio Natta (1903–1979) produced polypropylene in Milan and filed a patent application for it. In 1963, Karl Ziegler and Giulio Natta received the Nobel Prize for their “discoveries in the field of the chemistry and technology of high polymers.”
Structure and Properties
The chemically correct name for polypropylene is polypropene; the abbreviation PP is also commonly used.
In polypropylene, the methyl side group can be arranged in different ways. The arrangement of the methyl side group is described by tacticity: in isotactic PP, all methyl groups are located on one side of the polymer chain; in syndiotactic PP, the methyl groups alternate on both sides; and in atactic PP, they are randomly and irregularly distributed on both sides[1].
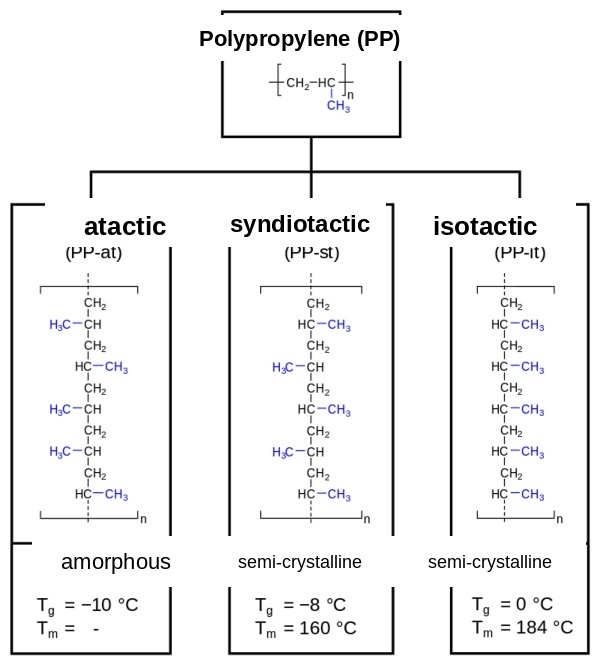
The arrangement of the methyl side group influences crystallinity and material properties. As crystallinity increases, the glass transition temperature, melting point, hardness, strength, and stiffness increase. With 70–80%[2], isotactic PP has the highest degree of crystallization among the three PP stereoisomers and has higher strength and stiffness compared with high-density polyethylene. The methyl groups arranged on one side force the polymer chain into a helical form. In syndiotactic PP, the degree of crystallization is 30–40%[2]. It is transparent and impact-resistant, but less stiff than isotactic PP. Atactic PP is amorphous and tacky at room temperature. For industrial applications, isotactic polypropylene is by far the most important.
Polypropylene has a density of less than 1 g/cm3, making it one of the “lightweight” plastics. At the same time, it is among the hardest plastics. With a glass transition temperature between +0 °C and -10 °C, PP becomes brittle below +0 °C. The maximum continuous service temperature is around +110 °C.
In addition, polypropylene is resistant to acids, alkalis, aliphatic hydrocarbons, alcohols, ketones, and water, which makes this plastic well suited for use in chemical laboratories and for laboratory equipment. It is attacked by oxidizing acids such as nitric acid and concentrated sulfuric acid, as well as by aromatic, cyclic, and halogenated hydrocarbons. Polypropylene is also not resistant to UV and X-ray radiation.


With a specific electrical resistivity >1016 Ω·cm and a dielectric strength of up to 50 kV/mm, this plastic is an insulator; its electrical properties are comparable to those of ceramics. PP has higher stiffness, hardness, and strength than polyethylene and also offers good barrier properties against moisture and fats. Like polyethylene, polypropylene does not contain plasticizers.
Industrial Production of Polypropylene
Polypropylene is a thermoplastic and belongs to the polyolefin group. The starting material is the monomer propene, which forms polypropylene at room temperature through radical polymerization in the presence of Ziegler–Natta catalysts. Ziegler–Natta catalysts are a combination of two metal compounds that create a catalytically active center to which the propene monomer attaches, allowing the polymer chain to begin growing. The choice of catalyst determines the position of the methyl side group. When triethylaluminum and titanium trichloride are used as catalysts, isotactic PP is obtained. To produce syndiotactic PP, diethylaluminum chloride and vanadium tetrachloride are used.
In addition to polypropylene—also referred to as polypropylene homopolymer (PP-H)—there are other polymers known as copolymers, which contain monomers other than propene. The most common is the ethylene–propylene copolymer, which is produced from the monomers ethene and propene, likewise in the presence of Ziegler–Natta catalysts.
Processing
PP plastics are available for further processing as uncolored and colored granules or powders. Uncolored, the material is gray.
In addition, the plastic can be compounded with fillers depending on application requirements. Adding mineral fillers such as talc, mineral fibers, glass fibers, or carbon fibers can improve hardness, strength, and stiffness. If ceramic fillers such as magnesium oxide, aluminum oxide, and aluminum nitride are added, a thermally conductive and electrically insulating plastic is obtained. By adding graphite or metallic powders such as copper and aluminum, the plastic becomes thermally and electrically conductive.
Films, fibers, and foams can be produced from polypropylene. With a density of 0.9 g/cm3, PP fibers are the lightest synthetic fibers and are used in the textile industry and in construction.

For films, a distinction is made between “oriented” PP (oriented PP, OPP) and “cast” PP (cast PP, CPP). “Oriented” PP is stretched in the longitudinal direction during production by rolling, which makes the film stiff and transparent. If it is then also stretched in the transverse direction, “biaxially oriented” PP (BOPP) is obtained. “Biaxially oriented” PP films are characterized by tear resistance, impact resistance, and puncture resistance. If an additional acrylic coating is applied, the material is called OPPA. “Cast” PP is not stretched and is less transparent and more elastic than OPP.
Foamed PP is referred to as “expanded” PP (EPP) or “porous expanded” PP (PEPP). It consists of 75 to 95% air, has a density between 20 and 250 kg/m3, and shows very good thermal insulation behavior, high dimensional stability, and higher impact and compressive strength than compact PP. Compared with a compact plastic, only about 10% material is needed, which makes EPP a resource-saving material.
Application Examples – From Textile Fiber to Beaker
Since polypropylene was first produced on a large industrial scale, production volume has increased rapidly: 200 tons of PP were produced in 1955, and by 2012 production volume had already reached 54 million tons. In industrialized countries, per-capita consumption is 18 kg/year, while in developing countries it is only 2 kg/year[3]. Like polyethylene, the cost of one kilogram of granules is around 1 euro/kg. Measured by revenue, polypropylene is the second most important commodity plastic worldwide after polyethylene and is used in numerous applications.
In the automotive industry, polypropylene is used as a material for dashboards, bumpers, air filter and headlight housings, spoilers, seat covers, child seats, and crash absorbers.


In construction, PP is used in many different areas. For example, this plastic can be found in fittings and pipelines for drinking-water installations, in air-conditioning and ventilation systems, as an insulating material, in furniture hinges, and in artificial turf. When concrete is mixed with PP fibers, frost resistance, abrasion resistance, compressive strength, and gas permeability increase significantly. In the event of a fire, the fibers decompose, forming microcracks that reduce internal stresses in the concrete and allow the resulting gases to escape[4,5].
Polypropylene is a component of the functional yarn Polycolon®, which is processed into various textiles. It is the lightest of the commercially available fibers, is skin-friendly, and has a high surface tension. This means it does not absorb water and sweat. For this reason, the functional yarn is often used to manufacture sportswear and outdoor clothing. It is also used in home textiles, carpets, ropes, and fishing nets.
In the chemical and pharmaceutical industries, laboratory containers such as measuring beakers, Erlenmeyer flasks, volumetric flasks, and evaporating dishes, plastic hoses, laboratory sinks, and disposable laboratory items such as syringes, spatulas and plastic containers are made from polypropylene.

In the household, many items are made of polypropylene: yogurt and margarine cups, bottle caps, plastic containers for detergents, housings for small electrical appliances such as coffee machines or kettles, and molded parts in household appliances such as washing machines or tumble dryers. Films made of CPP and OPP are used in packaging for vegetables and sweets. EPP is used to make thermal boxes, furniture, bicycle helmets, and model airplanes[6].
Recycling
Polypropylene is a highly versatile plastic that can be used in many different areas due to its properties. PP is easily recyclable, provided that single-grade purity is ensured. Products made from polypropylene can be identified by recycling code 5.
It can be processed for recycling and remelted into new products. When recycled material is used in the production of polypropylene, the energy consumption required for production can be significantly reduced. However, products made from recycled material may not be used in the food industry.
The incineration of polypropylene is unproblematic, as only water and carbon dioxide are produced. PP has a high calorific value, comparable to that of coal or oil.
According to the American Chemistry Council (ACC), the trade association of the chemical industry in the United States, the recycling rate of polypropylene in 2017 was less than 1%, although demand for recycled material is much greater[7]. Procter & Gamble developed a recycling process for polypropylene that yields high-purity recyclate. In cooperation with the U.S. company PureCycle Technologies Inc., a production facility was planned in Ohio in 2018 that was scheduled to produce 48,000 metric tons of recycled polypropylene from 54,000 metric tons of post-consumer polypropylene starting in 2021[8]. The plan also included applying for approval from the U.S. Food and Drug Administration (FDA), so that the recycled PP can also be used in the food industry.
Sources: 1) https://en.wikipedia.org/wiki/Polypropylene 2) https://en.wikipedia.org/wiki/Crystallization_of_polymers 3) https://www.kunststoffe.de/a/grundlagenartikel/marktentwicklung-polypropylen-pp-264429 4) https://opus4.kobv.de/opus4-bam/frontdoor/index/index/docId/35734 5) https://www.beton.wiki/index.php?title=Kunststofffaserbeton 6) https://epp-forum.com/einsatzgebiete 7) https://www.thebalancesmb.com/an-overview-of-polypropylene-recycling-2877863 8) https://purecycletech.wpengine.com/2019/09/successful-run-of-feedstock-evaluation-unit/
Image sources: Title image | © lukasvetic – stock.adobe.com Tacticity of polypropylene | © Minihaa, CC0, https://commons.wikimedia.org/w/index.php?curid=47058395 Colorfully dyed PP granules | © Emilia – stock.adobe.com Plastic recycling symbol | © Tomia, CC BY 2.5, https://commons.wikimedia.org/w/index.php?curid=591691
 Reichelt Chemietechnik Magazine
Reichelt Chemietechnik Magazine
